That’s because Burt was the subject of a 2016 warning letter from the Food and Drug Administration, which oversees clinical trials. “When you use it in the right group of patients with MS, you get these really gratifying results.”īut some researchers are also questioning the integrity of the study’s findings. “I do think it’s going to change the natural history of MS,” said Northwestern University stem cell researcher Richard Burt, the study’s lead author and Loy’s doctor. The small trial appears to provide more evidence that chemo and a stem cell transplant can help some people with MS. The rest saw their quality of life and disability improve. 
But for the 55 (including Loy) who received the chemo and stem cell transplant, only three got worse. The results of the trial appeared on January 15 in the journal JAMA : Among the 55 patients in the control group who took medication, 34 saw their disease worsen. Loy got it as part of the first randomized trial comparing the outcomes of patients with what’s called “relapsing remitting” MS who received the treatment to patients who took standard MS medications. The treatment is an experimental chemotherapy and hematopoietic stem cell transplant. (Heads up: You might be asked to sign in to Google first.) Become a member of the Vox Video Lab on YouTube today. “It sounds so dramatic, but gave me my life back,” she said. Her only lingering symptom is some mild nerve pain from time to time. She runs half-marathons and plays soccer with her son, who is 10. Nearly a dozen years on, Loy is now back to working full time as a teacher in the radiology program at the University of Alaska. It was around then that she decided to travel from her home in Anchorage, Alaska, to Chicago to inquire about a new treatment she’d heard about at a Seattle hospital. After she gave birth to a son a year later, in 2008, the symptoms worsened. Her hands and legs felt numb all the time, her bladder always felt full, and she had to rely on a cane to walk for more than 10 minutes. The story has been heavily updated with information about the letter and the perspectives of bioethicists and health law experts.īy the time Amanda Loy turned 28, her multiple sclerosis had progressed to the point that she could no longer work full time. "Hastening the return of the body’s white blood cells can reduce the possibility of serious or overwhelming infection associated with stem cell transplantation.Editor’s note 1/23/19: After this piece was published on January 15, a reader informed us that the lead author in the study described below had received a warning letter from the Food and Drug Administration in 2016. "Today's approval is an important advance in cell therapy treatment in patients with blood cancers," Peter Marks, M.D., Ph.D., director of the FDA Center for Biologics Evaluation and Research, said in a statement. 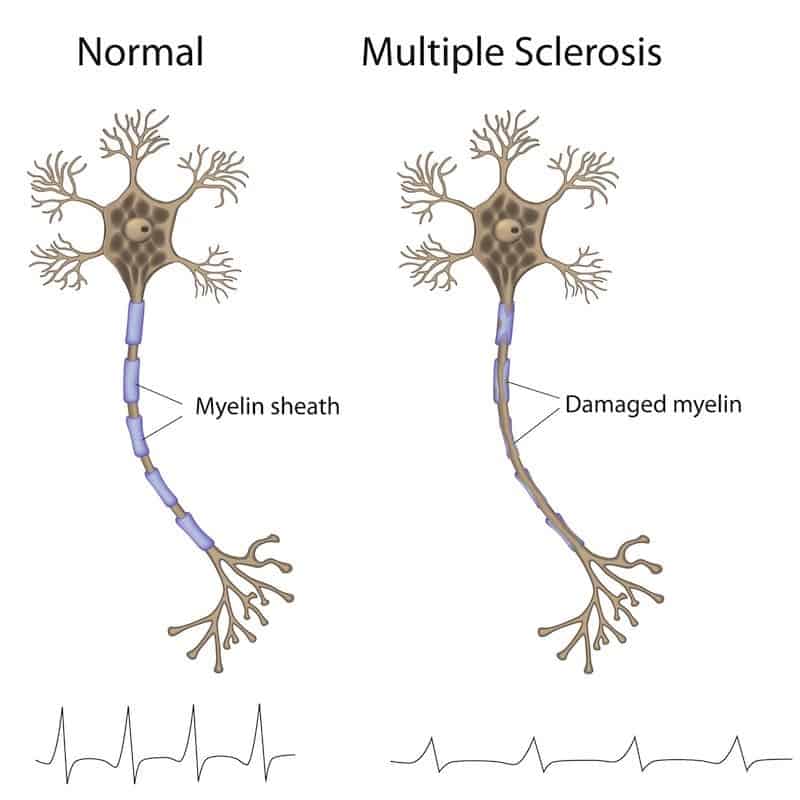
Within 100 days of transplantation, bacterial or fungal infections were seen in 39 percent of those receiving Omisirge versus 60 percent who received umbilical cord blood. Among patients randomly assigned to Omisirge, 87 percent achieved neutrophil recovery with a median of 12 days following treatment versus 83 percent of participants randomly assigned to receive umbilical cord blood transplantation (median 22 days for neutrophil recovery). The approval was based on data from a randomized, multicenter study comparing transplantation of Omisirge to transplantation of umbilical cord blood, in 125 patients with blood cancer (ages, 12 and 65 years). The treatment is composed of human donor stem cells from umbilical cord blood that are processed and cultured with nicotinamide. Omisirge is administered as a single, patient-specific intravenous dose for blood cancer patients ≥12 years to hasten recovery of neutrophils. Food and Drug Administration approved Omisirge (omidubicel-onlv), a substantially modified allogeneic cord blood-based cell therapy, to cut the risk of infection in patients with blood cancer following a myeloablative treatment, such as radiation or chemotherapy. FDA Approves Cord Blood Stem Cell Product, Omisirge, for Blood Cancer Patients
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
